The PI3K pathway is the most frequently activated pathway in cancer. PI3Ks, or PI 3-kinases, generate the inositol lipid PIP3 in the plasma membrane. This lipid molecule then binds to and activates proteins that will initiate signaling cascades in the cell, activating various cellular processes. Chief amongst these is the activation of cell growth, proliferation and migration. This happens when the PI3Ks are activated downstream of growth factor receptors. Or, when mutations cause the cell to overproduce too much PIP3 in the resting state – in which case, growth, proliferation and migration happens in cells when it shouldn’t. This process is what we call cancer.
Why is this pathway so commonly found in cancer (more than half of all tumors)? Well, probably because there are multiple ways to overproduce PIP3. Firstly, the genes encoding PI3K enzymes can acquire activating mutations – i.e., they are oncogenes. Secondly, an enzyme that usually degrades PIP3 to switch off signaling can become mutated so it doesn’t work anymore – i.e., it is a tumor suppressor.
What can be done about this? Well, lots of big pharma have produced drugs that block the PI3Ks. The problem is, PI3Ks don’t just drive cancer proliferation – they do lots of other jobs in the body, such as controlling the cells’ response to insulin and activating the immune system. So these drugs can be very bad for us. Clearly, we need a more nuanced approach.
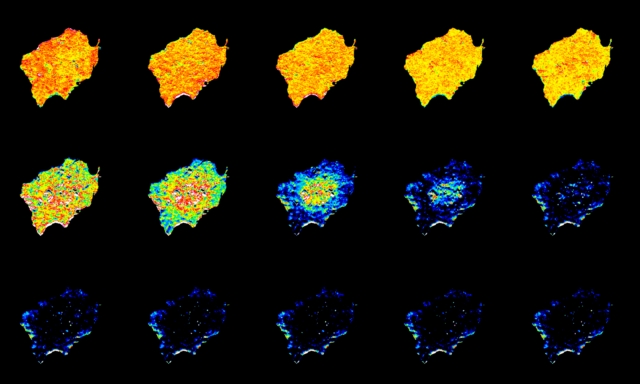
In the Hammond lab, we are studying a side-branch of the PI3K pathway. The lipid PIP3 is often converted into a side product, PI(3,4)P2. This lipid isn’t always made to a large degree – but it is downstream of many of the growth factor that cause the growth, proliferation and migration process that are associated with cancer. So, we are trying to focus in on how and where this lipid is produced, and what exactly it does. To this end, we’ve been developing new biosensors to track the lipid, and new opto- and chemical genetics tools to manipulate it. The image above shows a novel biosensor being shed from the membrane when we use chemical genetics to acutely destroy the PI(3,4)P2.
In the long term, we hope that the basic knowledge we can uncover about the function of this lipid will help the drug companies develop more tailored medicines targeting the unwanted signaling events happening specifically inside cancer cells.
